The following article is intended to describe an important part of the “lateral flow-world”: the combination of nucleic acid amplification and lateral flow.

More than just a Platform for Nucleic Acid Amplification and Lateral Flow
Milenia HybriDetect is a universal Lateral Flow Development Platform that is cited in more than 145 publications. The overwhelming majority of these peer reviewed papers decribe quick and easy identification of DNA-amplification products. This methodological combination of DNA-/ RNA-amplification and lateral flow allows the decoupling of complex methods from specialized laboratories and thus the development of field deployable tests. Although the following sections refer specifically to nucleic acid amplification techniques, the Milenia HybriDetect can be more than just an amplicon detection format.
Learn more about Lateral Flow Basics, Milenia HybriDetect Composition and Detection Principles
How to handle the Milenia HybriDetect
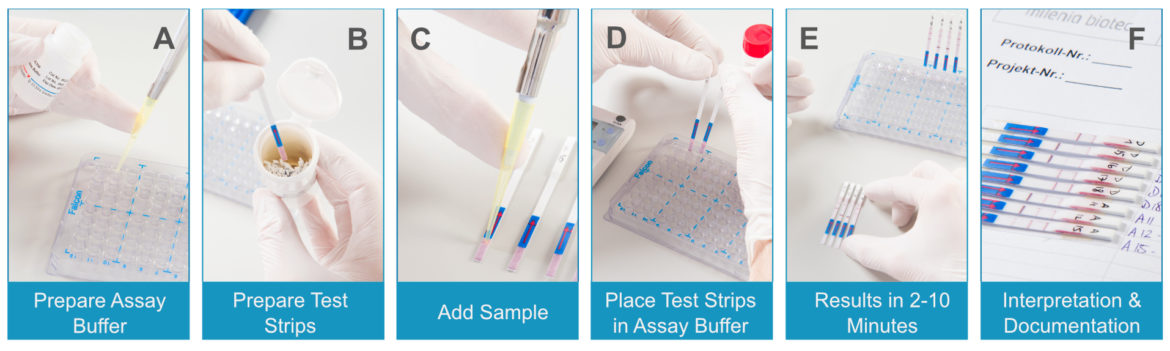
The general processing of the Lateral Flow Analysis remains almost the same regardless of which application is combined with this procedure. The sample can be applied directly to the strip or into the assay buffer (Fig.1, 1-3). The test strips are positioned vertically in a volume between 50 and 100 µL, which initiates the lateral flow (Fig.1, 4). Recognizable signals can appear after a few seconds. The results are usually interpreted after 2-10 minutes (Fig.1, 5).
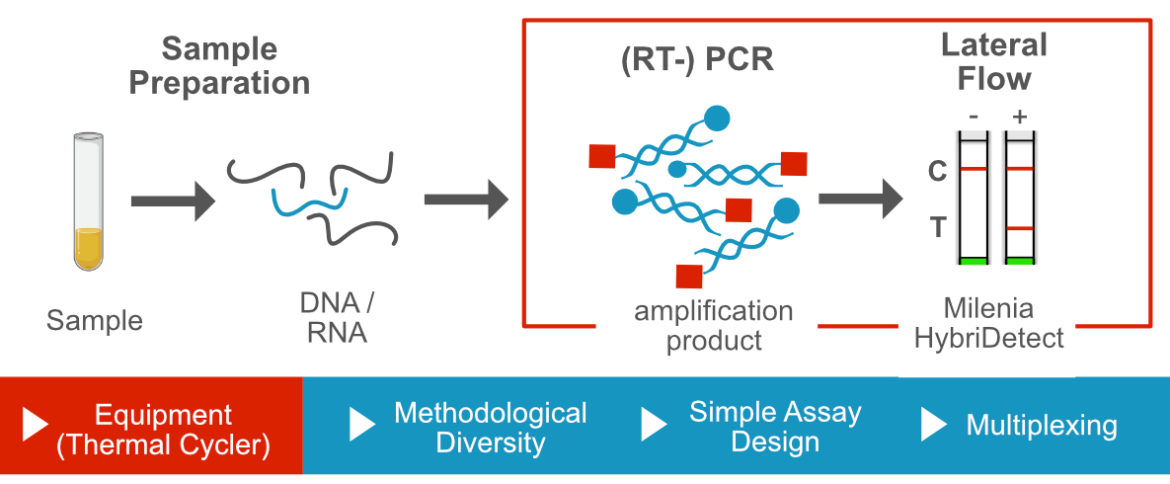
Polymerase Chain Reaction and Lateral Flow
The polymerase chain reaction is the frist described, best understood and most important DNA-amplification method worldwide. Ultimately, all types of DNA-amplification techniques are based on this technology. Nevertheless, PCR is a relatively underrepresented technique for point-of-care applications. PCR is often described as laborious, expensive and time intensive. Furthermore, PCR needs special equipment and highly trained personnel. These arguments are admittedly one-sided and neglect the important advantages of this method. The simple assay design, the methodological versatility, the multiplex compatibility, the development in reagents and the large number of already established PCR-variations make this method an absolutely underrated technique in the field of point-of-care testing.
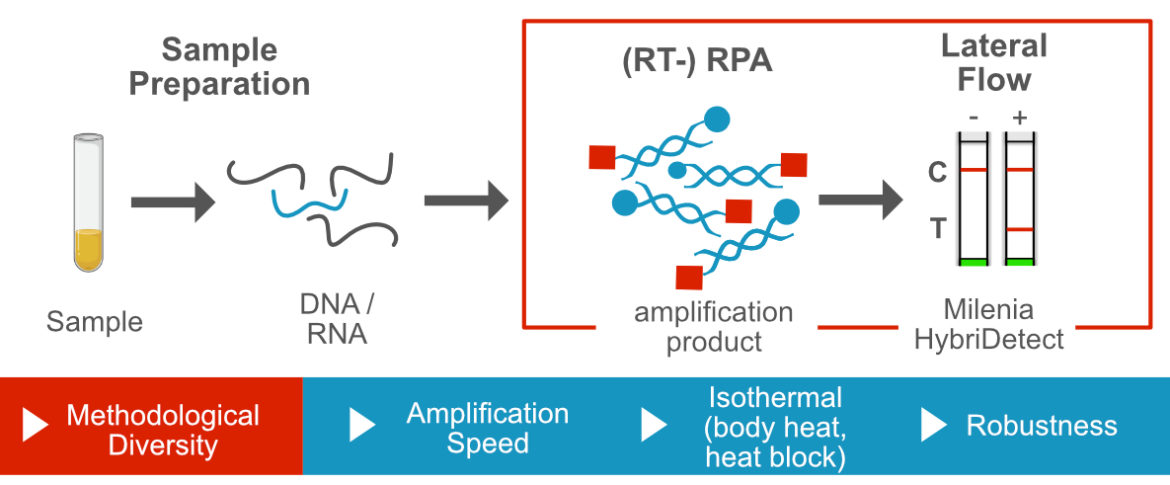
Recombinase Polymerase Amplification and Lateral Flow
The Recombinase Polymerase Amplification (RPA) uses beside a polymerase with strong displacement activity special components (recombinase, ssDNA-binding protein, loading factor, crowding agent) to allow specific, isothermal amplification at lower temperatures between 37 and 42°C. The RPA is an extremely fast DNA-amplification technique, that is very robust against several inhibitors. Taken together, the RPA is one the most popular methods for fast, sensitive, specific and field-deployable DNA-amplification. The combination of the recombinase polymerase amplification and lateral flow based amplicon detection allows the development of almost equipment free rapid tests that are particularly useful in applications under low-resource-settings.
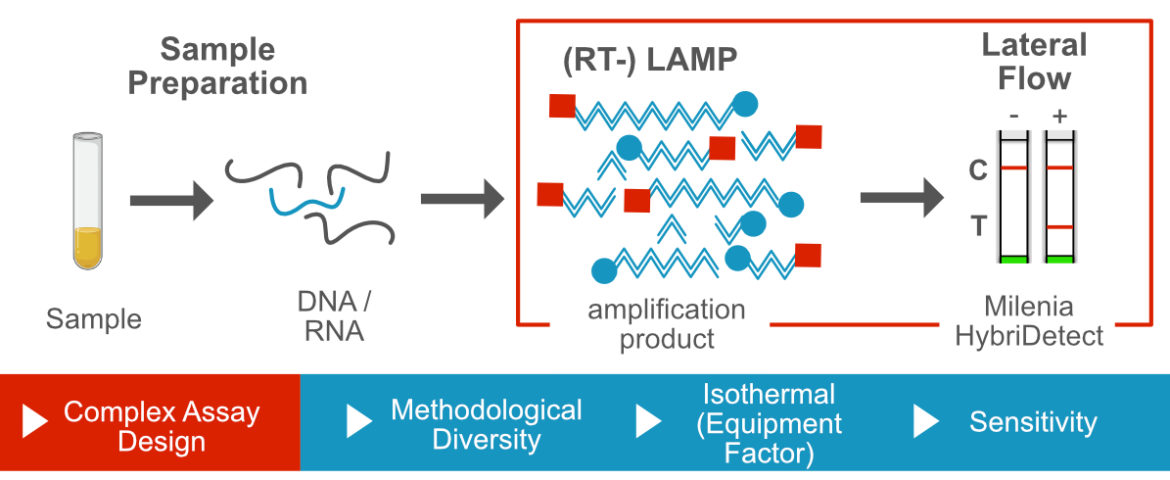
Loop mediated isothermal Amplification and Lateral Flow
The loop mediated isothermal amplification (LAMP) is one the most frequently used isothermal DNA-amplification methods. The use of multiple primers in combination with a DNA polymerase with strong strand displacement activity and constant temperatures of 60-65°C allow rapid and sensitive DNA-detection. One characteristic of the LAMP is the methodological diversity allowing extremly sensitive assays or advanced specificity (e.g. SNP-detection). The combination of multiplex LAMP with a lateral flow-based amplicon detection is one the most promising tools in POC-DNA-diagnostics so far. Learn more about LAMP and Lateral Flow
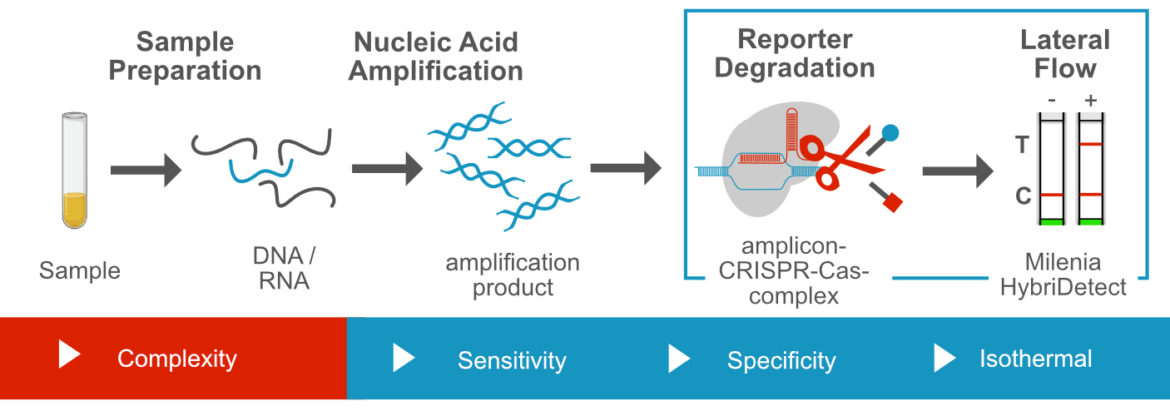
CRISPR/Cas-dependent Nuclease Assays and Lateral Flow
In most cases these methods combine isothermal amplification methods, like LAMP, RPA or NASBA with a specific CRISPR/Cas based nuclease assay. Two consecutive enhancer reactions result in ultrasensitive diagnostic applications that are particularly interesting for detection of viral pathogens. The specific breakdown of dual labeled reporter is detectable with the Milenia HybriDetect. The combination of sensitivity and specificity of CRISPR / Cas-dependent assays makes this comparatively new approach to one the most exiting topics in the field of molecular diagnostics.
- CRIPSR/Cas 12 – based detection, e.g. DETECTR
- CRISPR/Cas 13 – based detection, e.g. SHERLOCK, INSPECTR
- CRISPR/Cas 14 – based detection (future application)
- CRISPR/Cas 3 – based detection (principle shown)
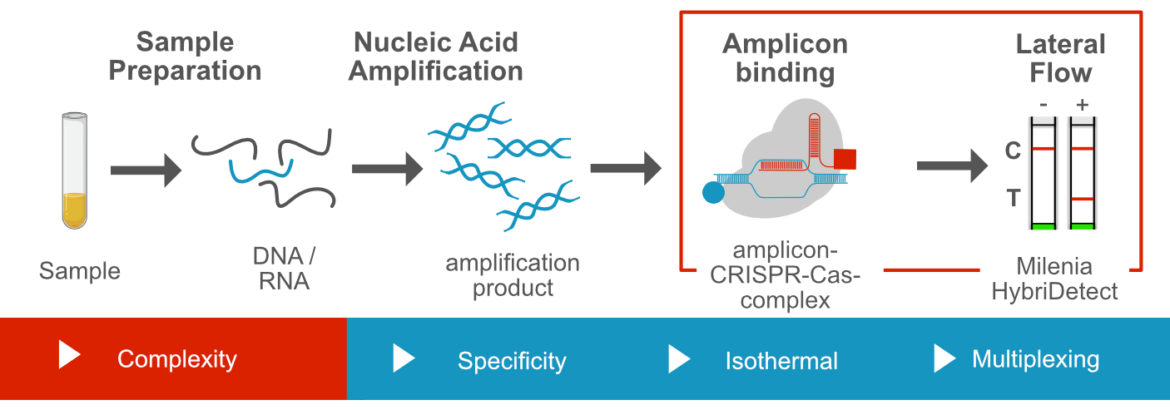
CRISPR/Cas dependent „Amplicon Binding Assays“ and Lateral Flow
In most cases these methods combine DNA amplification with a CRISPR/Cas-based amplicon „binding assay“. The benefit of this methodological combination is the impressive specificity of the CRISPR/Cas-mediated amplicon „recognition“. A striking example is a preprint, presented in february by Kumar and colleages. Cas9 from Fransicella novidica is used for identification of SARS-CoV-2 mutant strains due to specific amplicon recognition.
More Promising Tools for Nucleic Acid Amplification and Lateral Flow
Today, there are many different variations of isothermal techniques that combine nucleic acid amplification and lateral flow. The focus in the literature is very much on LAMP and RPA. But other classic isothermal techniques such as helicase dependent amplification (HDA), rolling circle amplification (RCA), cross priming amplification (CPA) or strand displacement amplification (SDA) are perfectly compatible with a simple lateral flow readout. But also less frequently used methods such as polymerase spiral amplification (PSA) or nucleic acid sequence based amplification (NASBA) hold great potential in the context of nucleic acid based rapid testing.
It gets particularly interesting if innovative molecular tools like the CRIPSR-Cas „machinery“, additional DNA-modifiying enzyms or DNAzymes are combined with existing DNA-or RNA-amplification methods. A good and comprehensive knowledge of the toolbox is the basis for the development of innovative assays formats & nucleic acid detection platforms. The following image summarizes important parts of this toolbox for nucleic acid lateral flow immunoassays.

Outlook – New Methods, Alternative Techniques, Future Trends and Applications
Innovative molecular detection techniques such as SHERLOCK, FELUDA or LEOPARD rely on the use of the CRISPR / Cas technology. Regardless of the future importance of these new tools, they clearly show what the challenges of a new generation of molecular diagnostics are. On the one hand, it is about finding ways to take laboratory analysis to a new level. Especially high throughput analyzes that provide much more information than a simple “positive” or “negative” are certainly the focus of modern molecular analytics. On the other hand, the SARS-CoV-2 pandemic has clearly shown the importance of a simple, robust and yet sensitive and precise analysis under low resource settings. Preventive “at-home-testing” in the SARS-CoV-2 pandemic has shifted the awareness of diagnostics outside of specialized laboratories. If you take a look at the initial publications of the above-mentioned methods, it becomes apparent that point-of-care analysis plays a particularly important role.
Because sensitive, molecular diagnostics must also work outside of specialized laboratories. This is exactly where the compatibility with extremely simple, robust, instrument-free downstream analytics such as the lateral flow technology is one of the keys to modern POC diagnostics.
The third and last part of this blog series is dedicated to give more information about existing fields of application and thus highlights more trends and challenges of nucleic acid lateral flow immunoassays.




